  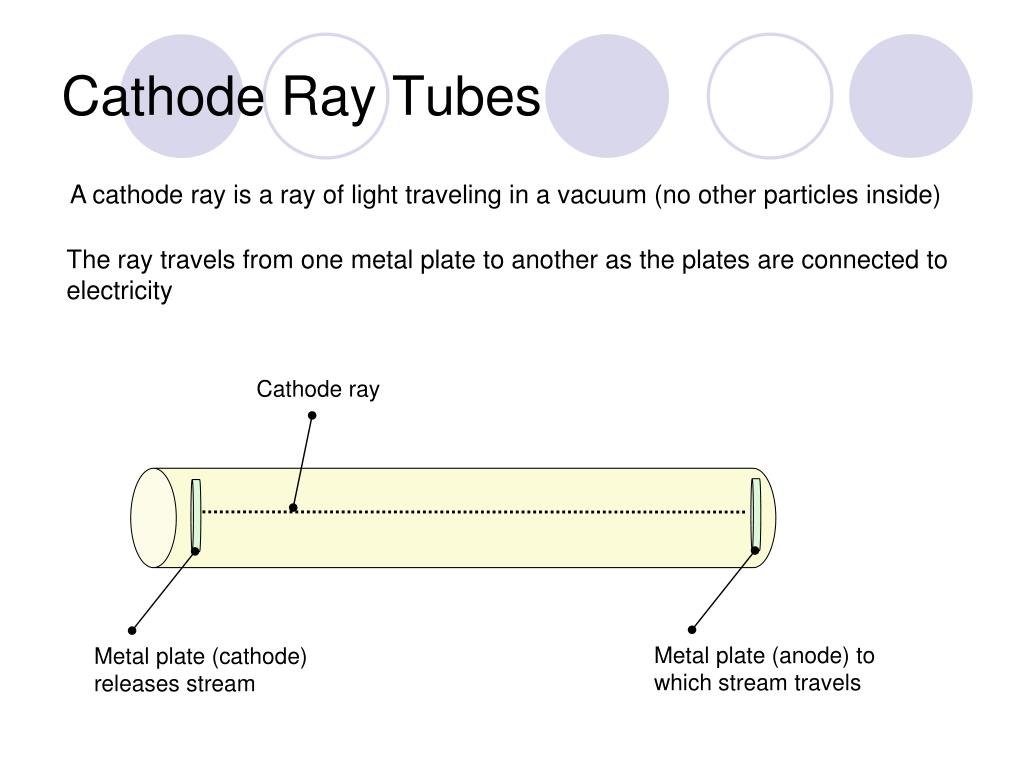
In the early experimental cold cathode vacuum tubes in which cathode rays were discovered, called Crookes tubes, this was done by using a high electrical potential of thousands of volts between the anode and the cathode to ionize the residual gas atoms in the tube. To release electrons into the tube, they first must be detached from the atoms of the cathode. The Maltese cross has no external electrical connection.Ĭathode rays are so named because they are emitted by the negative electrode, or cathode, in a vacuum tube. Cathode-ray tubes (CRTs) use a focused beam of electrons deflected by electric or magnetic fields to render an image on a screen.Ī diagram showing a Crookes tube connected to a high voltage supply. Thomson showed that cathode rays were composed of a previously unknown negatively charged particle, which was later named the electron. They were first observed in 1859 by German physicist Julius Plücker and Johann Wilhelm Hittorf, and were named in 1876 by Eugen Goldstein Kathodenstrahlen, or cathode rays. If an evacuated glass tube is equipped with two electrodes and a voltage is applied, glass behind the positive electrode is observed to glow, due to electrons emitted from the cathode (the electrode connected to the negative terminal of the voltage supply). Cathode rays are normally invisible in this Teltron tube demonstration, enough gas has been left in the tube for the gas atoms to luminesce when struck by the fast-moving electrons.Ĭathode rays or electron beam ( e-beam) are streams of electrons observed in discharge tubes. Also, thermionic emission allowed a much higher bound on the numbers of electrons produced, and hence lead to a drastic reduction in exposure times.A beam of cathode rays in a vacuum tube bent into a circle by a magnetic field generated by a Helmholtz coil. In fact, as the number of electrons produced depended on the current applied across the cathode filament, and the energy of the electrons on the tube voltage, the Coolidge tube made it possible to easily and independently vary the number of electrons (and hence the intensity of x-rays produced), and their energy (and hence the frequency of produced x-rays). 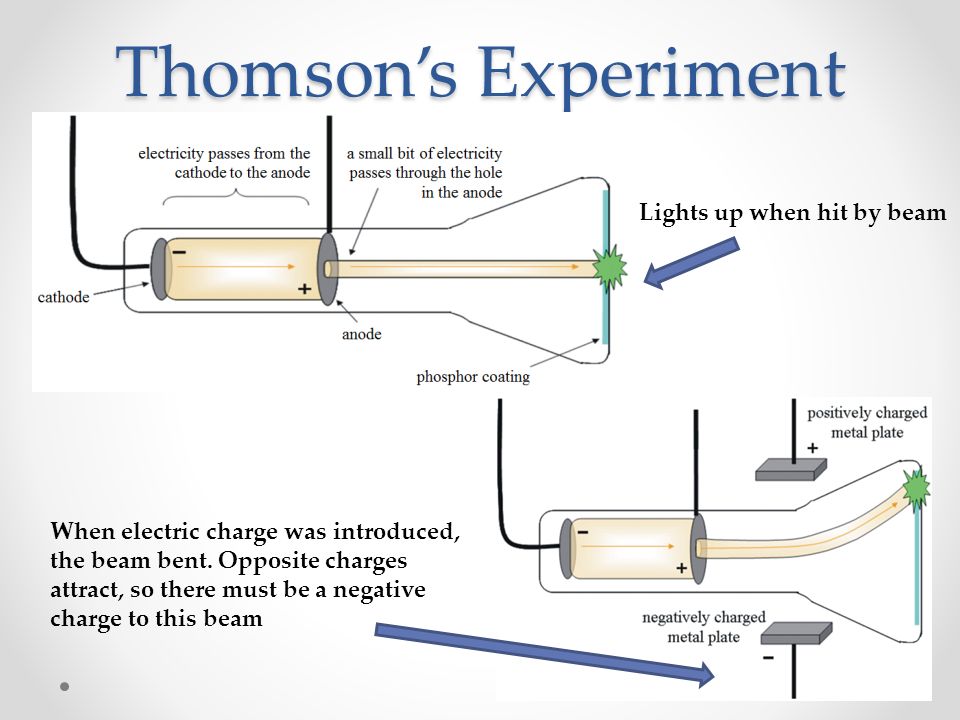
The Coolidge tube, using as it did thermionic emission to obtain a source of electrons, removed the dependency on residual gas for the number and energy of the electrons in the electron beam (an indeed in Coolidge tubes, almost all gas is removed). In addition, the number of electrons produced in this way, was, by today's standards extremely small, and hence the intensity of the produced x-rays very low, leading to very long exposure times. However, the number of electrons in a beam produced this way, and their energy upon collision with the anode, were both dependent on the gas pressure within the tube, which was rarely stable, and difficult to control. In this way, the required electron beam was built up with a kind of 'avalanche effect'. These were accelerated towards the anode, and collided with residual gas molecules purposefully left in the tube, ionizing the molecules and causing the ejection of more electrons. X-ray tubes previous to the Coolidge tube (known as Gas Tubes), relied for their electron source, on the tube voltage being strong enough to 'pull' electrons from the cathode. To prevent the electron beam from dispersing due to repulsive forces between the electrons, the cathode filament is surrounded by a metal focusing cup at a high negative potential, that has the effect of converging the beam to a relatively small focal area on the anode. Upon hitting the anode, the electrons are decelerated very rapidly, and shed their excess kinetic energy mostly as heat, and partly as x-ray radiation. The electrons are then accelerated towards the tube anode by the strong tube voltage. This causes the filament to emit electrons at a rate dependent on the temperature of the filament. The Coolidge tube was the first type of practical x-ray tube to employ the principle of thermionic emission.Ī tungsten filament is used as the tube cathode, and during operation is heated to incandescence by passing a current through it. Coolidge, is the forerunner of all the types of x-ray tubes in common use today. The Coolidge Tube, first produced in 1913 by W. Spherical glass bulb (purple from radiation damage) with cylindrical stems carrying the electrodes (cathode end broken). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
